 Atlanta, GA – Centers for Disease Control and Prevention (CDC) is aware that New England Compounding Center has voluntarily expanded its recall to include all products currently in circulation that were compounded at and distributed from its facility in Framingham, Massachusetts.
Atlanta, GA – Centers for Disease Control and Prevention (CDC) is aware that New England Compounding Center has voluntarily expanded its recall to include all products currently in circulation that were compounded at and distributed from its facility in Framingham, Massachusetts.
The Fungal Meningitis outbreak now covers 9 States and the case count has grown from 64 to 91. Tennessee has 3 new cases.
Clinicians should actively contact patients who have received medicines associated with three lots of preservative-free methylprednisolone acetate (80mg/ml) recalled on September 26th.
The potentially contaminated injections were given starting May 21st, 2012. Symptoms that should prompt diagnostic evaluation include: fever, new or worsening headache, neck stiffness, sensitivity to light, new weakness or numbness, increasing pain, redness or swelling of the injection site.
At A Glance
- Status: Ongoing Investigation
- Infection: Fungal Meningitis
- Facility Type: Outpatient Setting
- Case Count: 91
- States: 9
- Deaths: 7
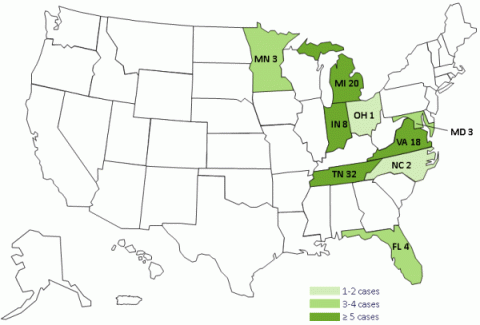
Cases By State
- Florida: 4 cases
- Indiana: 8 cases
- Maryland: 3 cases, including 1 death
- Michigan: 20 cases, including 2 deaths
- Minnesota: 3 case
- North Carolina: 2 cases
- Ohio: 1 case
- Tennessee: 32 cases, including 3 deaths
- Virginia: 18 cases, including 1 death
Clinician Guidance
October 6th, 2012 8:00pm ET
- CDC Health Alert Network (HAN): Meningitis and Stroke Associated with Potentially Contaminated Product
- Interim Treatment Guidance for Central Nervous System and/or Parameningeal Infections Associated with Injection of Potentially Contaminated Steroid Products New: Oct 6th, 2012
- Case Definitions: Multistate Outbreak Associated with Injection of Potentially Contaminated Steroid Product New: Oct 6th, 2012
- Instructions for Clinical Teams Regarding Diagnostic Testing and Specimen Submission to CDC – Outbreak Associated With Injection of Potentially Contaminated Steroid Products New: Oct 6th, 2012
- Frequently Asked Questions For Clinicians: Multistate Meningitis Outbreak Investigation Updated: Oct 7th, 2012
- Role of antifungal prophylaxis in asymptomatic patients New: Oct 6th, 2012
- Lumbar puncture in asymptomatic patients New: Oct 6th, 2012
Check the CDC website’s Clinician Guidance page for further updates.
For more on the story see:
Centers for Disease Control and Prevention coordinates Multistate Meningitis Outbreak Investigation


